

Product Name : Minoxidil
CAS : 38304-91-5
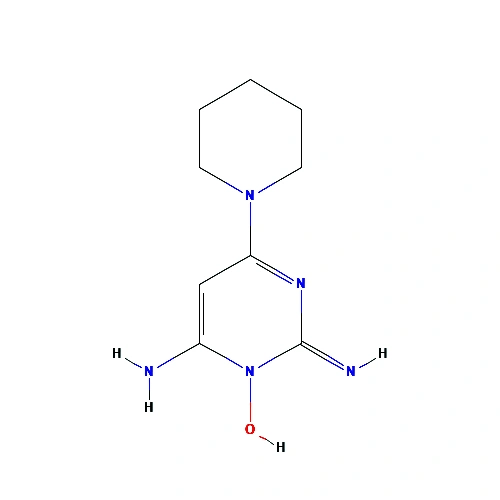
Molecular Formula : C9H15N5O
Molecular Weight : 209.25 g/mol
Pharmaceutical Grade : EP / IP / USP
Therapeutic Category : Antihypertensive, Antialopecia Agent
Global calcium is one of the leading manufacturers and exporters of minoxidil CAS no. 38304-91-5, API, (Active Pharmaceutical Ingredient). We support the customers with exhaustive documentation. As a USDMF & EU-GMP certified global company and an established hallmark for pharmaceutical standards, global calcium has stood the test of time since its inception in 1979.
As manufacturer of minoxidil we hereby state the following facts about the drug:
Androgenic alopecia: Androgenic alopecia (AGA) is a partial or complete loss of hair that occurs in a progressive pattern in genetically predisposed individuals.
Minoxidil induces cell growth factors on the plasma membrane of dermal papilla cells and also inhibits apoptosis of hair matrix cells.
It dilates hair follicle arteries and increases blood flow in dermal on the plasma membrane of vascular smooth muscle cells.
Alopecia areata: It is itchy non-scarring alopecia to complete scalp (alopecia totalis) and body (alopecia universalis) hair loss. Minoxidil is occasionally used off-label as monotherapy or in combination with other treatments.
Chronic telogen effluvium: It is a common non-scarring alopecia characterized by excessive telogen hair shedding triggered by stressful events such as pregnancy, a major illness, and surgery. Chronic telogen effluvium (CTE) is defined as hair loss persisting over 6 months. Minoxidil is proven to be highly effective in the treatment.
Scarring alopecia: In scarring alopecia, medical treatment should be initiated as early as possible with minoxidil. The aim of the treatment is to preserve the remaining hair follicles and halt disease progression.
Chemotherapy-induced alopecia: Hair loss is one of the common side effects of chemotherapy. A randomized controlled trial showed that 1 mL 2% MS applied to the entire scalp twice daily shortened the duration of alopecia by approximately 50 days in patients.
Hair shaft disorders: Minoxidil has been used to treat monilethrix, a rare autosomal dominant hair disorder manifested as fragile hair shafts with a regular beaded appearance.
Body hair enhancement: Topical minoxidil has been employed to stimulate hair growth on eyebrows and beard growth.
Global calcium is a leading manufacturer of this drug. We manufacture this pharmaceutical drug and make it available to domestic and overseas market